The structure of a folding intermediate provides insight into differences in immunoglobulin amyloidogenicity
02-Jul-2008
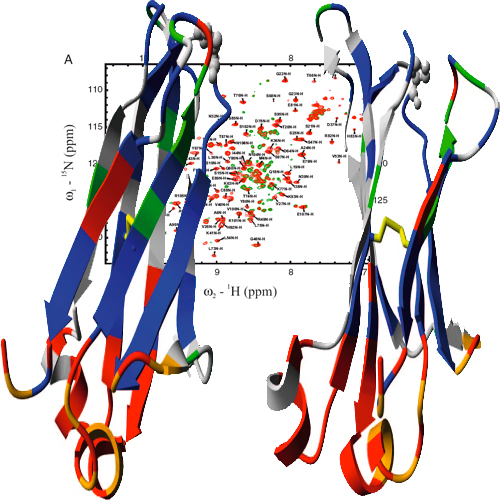
Folding intermediates play a key role in defining protein folding and assembly pathways as well as those of misfolding and aggregation. Yet, due to their transient nature, they are poorly accessible to high-resolution techniques. Here, we made use of the intrinsically slow folding reaction of an antibody domain to characterize its major folding intermediate in detail. Furthermore, by a single point mutation we were able to trap the intermediate in equilibrium and characterize it at atomic resolution. The intermediate exhibits the basic b-barrel topology, yet some strands are distorted. Surprisingly, two short strand-connecting helices conserved in constant antibody domains assume their completely native structure already in the intermediate, thus providing a scaffold for adjacent strands. By transplanting these helical elements into b2-microglobulin, a highly homologous member of the same superfamily, we drastically reduced its amyloidogenicity. Thus, minor structural differences in an intermediate can shape the folding landscape decisively to favor either folding or misfolding.