cis-Peptide Bonds: A Key for Intestinal Permeability of Peptides?
04-Sep-2015
Chem. Eur. J. 2015, 21, 15148 – 15152, DOI: 10.1002/chem.201501600
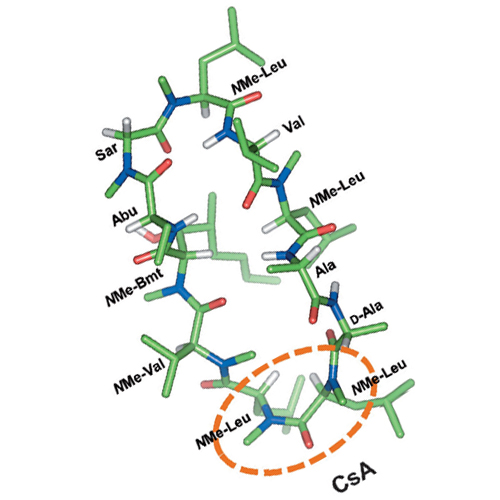
Recent structural studies on libraries of cyclic hexapeptides led to the identification of common backbone conformations that may be instrumental to the oral availability of peptides. Furthermore, the observation of differential Caco-2 permeabilities of enantiomeric pairs of some of these peptides strongly supports the concept of conformational specificity driven uptake and also suggests a pivotal role of carrier-mediated pathways for peptide transport, especially for scaffolds of polar nature. This work presents investigations on the Caco-2 and PAMPA permeability profiles of 13 selected N-methylated cyclic pentaalanine peptides derived from the basic cyclo(-D-Ala-Ala4-) template. These molecules generally showed moderate to low transport in intestinal epithelia with a few of them exhibiting a Caco-2 permeability equal to or slightly higher than that of mannitol, a marker for paracellular permeability. We identified that the majority of the permeable cyclic penta- and hexapeptides possess an N-methylated cis-peptide bond, a structural feature that is also present in the orally available peptides cyclosporine A and the tri-N-methylated analogue of the Veber–Hirschmann peptide. Based on these observations it appears that the presence of N-methylated cis-peptide bonds at certain locations may promote the intestinal permeability of peptides through a suitable conformational preorganization.