Photochromic Bis(thiophen-3-yl)maleimides Studied with Time-Resolved Spectroscopy
14-Jan-2009
The Journal of Physical Chemistry, 2009, 113, 6, 1033- 39 published on 14.01.2009
The Journal of Physical Chemistry, online article
The Journal of Physical Chemistry, online article
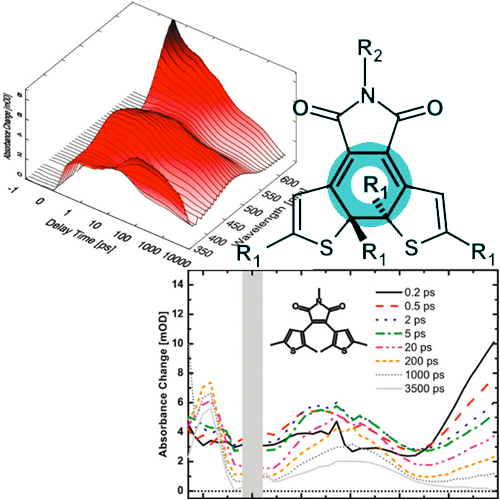
The dynamics of the ring-closure reaction of three different bis(thiophen-3-yl)maleimides are investigated using ultrafast spectroscopy in the visible range. The structures of the molecules differ with respect to substitution of the thiophene ring and the maleimide. The experiments reveal reaction kinetics which point to the population of an excited electronic state for several nanoseconds. In the case of completely unsubstituted thiophene rings, a long excited-state lifetime (biexponential decay with 3 and 15 ns) can be observed. The remaining ultrafast absorption transients of this molecule are due to relaxational processes on the excited electronic potential energy surface. The ring-closure reaction has a small yield (<1%) and does not show up in the ultrafast absorption experiments. A dimethyl substitution of the thiophene ring results in completely different behavior: after transients related to relaxation in the excited electronic state, one finds pronounced absorption transients with τ = 16 ps which represent the partial decay of the excited electronic state and the formation of the ring-closed isomer. Another fraction of the emitting excited electronic state decays again on the few nanosecond time scale. The experiments suggest that the open isomer of the dimethyl-substituted imides exists in two conformations.